 Direct-total bilirubin ratio was very variable with some 95th centiles >30%. The 95th centile for direct bilirubin was stable at ~25 µmol/L. 
The ratio between the two did not change significantly (-0.0006 to +0.0034). For each day of older age, total bilirubin fell by 3.72 µmol/L (95% CI 2.46 to 5.00) and direct bilirubin fell by 0.39 µmol/L (0.18 to 0.59). The association was assessed using linear regression analysis.ĭata were collected for 420 neonates (501 blood samples) across an age range of 10-70 days. We plotted centile charts for total, direct and direct-total bilirubin ratio levels against age at sampling. We performed a retrospective cohort analysis of split bilirubin levels, and subsequent follow-up, for all neonates initially assessed in our prolonged neonatal jaundice clinic over 2 years. 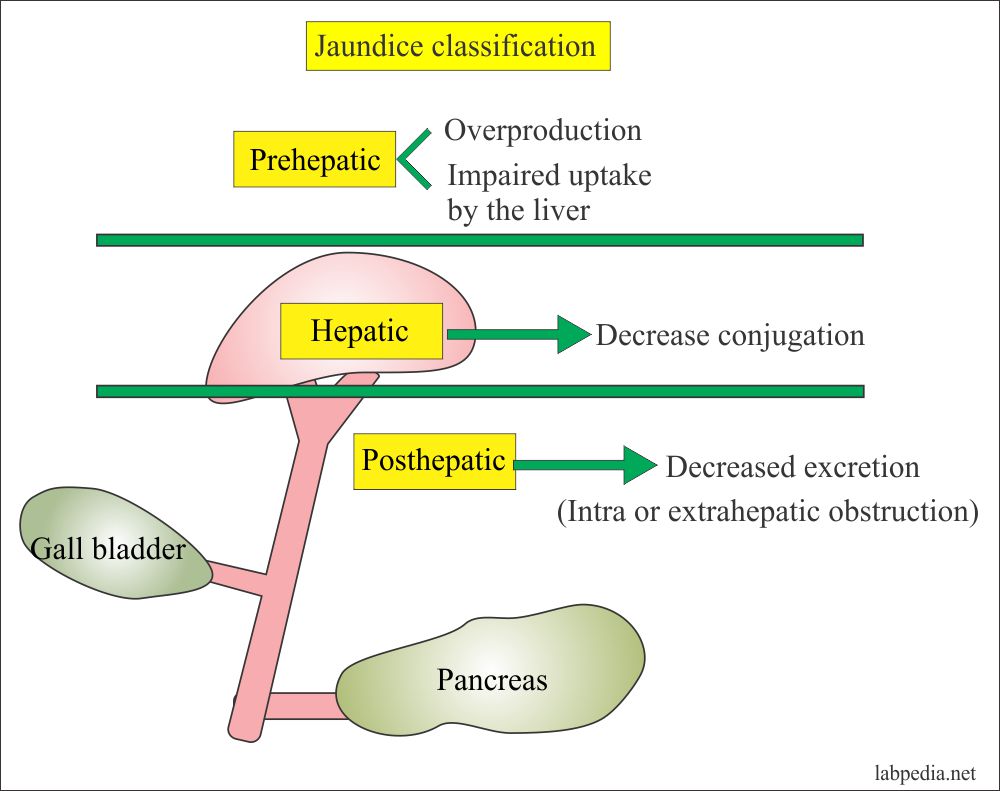 
Hence, we aimed to delineate direct bilirubin levels in disease-free neonates with prolonged jaundice. The direct (conjugated) bilirubin thresholds currently used are variable and poorly evidenced. Nothing in this guideline should be interpreted in a way that would be inconsistent with complying with those duties.Ĭommissioners and providers have a responsibility to promote an environmentally sustainable health and care system and should assess and reduce the environmental impact of implementing NICE recommendations wherever possible.Prolonged neonatal jaundice is common and usually benign however, assessment of bilirubin fractions is recommended to determine the need for further assessment for congenital liver disease, particularly biliary atresia. They should do so in the context of local and national priorities for funding and developing services, and in light of their duties to have due regard to the need to eliminate unlawful discrimination, to advance equality of opportunity and to reduce health inequalities. Local commissioners and providers of healthcare have a responsibility to enable the guideline to be applied when individual professionals and people using services wish to use it. It is not mandatory to apply the recommendations, and the guideline does not override the responsibility to make decisions appropriate to the circumstances of the individual, in consultation with them and their families and carers or guardian.Īll problems (adverse events) related to a medicine or medical device used for treatment or in a procedure should be reported to the Medicines and Healthcare products Regulatory Agency using the Yellow Card Scheme. When exercising their judgement, professionals and practitioners are expected to take this guideline fully into account, alongside the individual needs, preferences and values of their patients or the people using their service. The recommendations in this guideline represent the view of NICE, arrived at after careful consideration of the evidence available. For more information, see the surveillance decision. The update will focus on total serum bilirubin thresholds for starting phototherapy or exchange transfusion in term babies with neonatal hyperbilirubinaemia.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
